Getting started with Salmon
Unzip RAR files on your Mac from the Context menu. With WinZip for Mac 2.0 or later, you can right-click (or press CTRL and click) your saved RAR files and use the Context Menu to unzip them. Click on Services at the bottom of the Context menu, then choose Unzip. Buy WinZip for Mac. RNA-seq aligner. Contribute to alexdobin/STAR development by creating an account on GitHub. 64 bit Linux or Mac OS X. Download the latest release from.
https://builderrenew286.weebly.com/gear-watch-designer-download-mac.html. This brief tutorial will explain how you can get started using Salmon to quantify your RNA-seq data. This tutorial will walk you through installing salmon, building an index on a transcriptome, and then quantifying some RNA-seq samples for downstream processing.
Index of this tutorial:
Obtaining Salmon
Salmon is a free (both as in “free beer” and “free speech”) software tool for estimating transcript-level abundance from RNA-seq read data. It is developed openly on GitHub. You can visit Salmon’s GitHub page here, and check out the Salmon source code, feature requests, known issues etc.
The easiest way to install salmon is likely via bioconda.
Download youtube playlist mac os x. This will install the latest salmon in its own conda environment. The environment can then be activated via: https://sitefrench910.weebly.com/download-civilization-5-demo-mac.html.
Another way to get started with Salmon is to download the pre-compiled binaries for your platform from the releases page.
If you’ve downloaded a specific binary, you simply decompress it like so:
If you’ve downloaded a specific binary, you simply decompress it like so:
then, the binary will be located in the
bin directory inside of the uncompressed folder. You can either run salmon directly using the full path, or place it into your PATH variable for easier execution. The rest of the tutorial below will assume that you’ve placed the salmon executable in your path, so that simply running salmon will invoke the program. You can test that salmon is running on your system and get a list of available commands using the -h command; you should see output like the followingNote: OSX is frustratingly particular about how it looks for dynamic symbols in programs. If you’re on OSX and you’re getting an unresolved symbol error, you should run Salmon with the library directory in you
DYLD_FALLBACK_LIBRARY_PATH, like this:now, Salmon should find the appropriate symbols.
Download Rna Star For Macbook Pro
Obtaining a docker image for Salmon
Salmon is also available via Docker hub. You can obtain a docker image of salmon using the command:
Then, if you wish, you can follow the tuorital below using this contanerized version of Salmon.
Analyzing your RNA-seq data with Salmon
Obtaining a transcriptome and building an index
In order to quantify transcript-level abundances, Salmon requires a target transcriptome. This transcriptome is given to Salmon in the form of a (possibly compressed) multi-FASTA file, with each entry providing the sequence of a transcript1. For this example, we’ll be analyzing some Arabidopsis thaliana data, so we’ll download and index the A. thaliana transcriptome. First, create a directory where we’ll do our analysis, let’s call it
salmon_tutorial:Now, download the transcriptome:
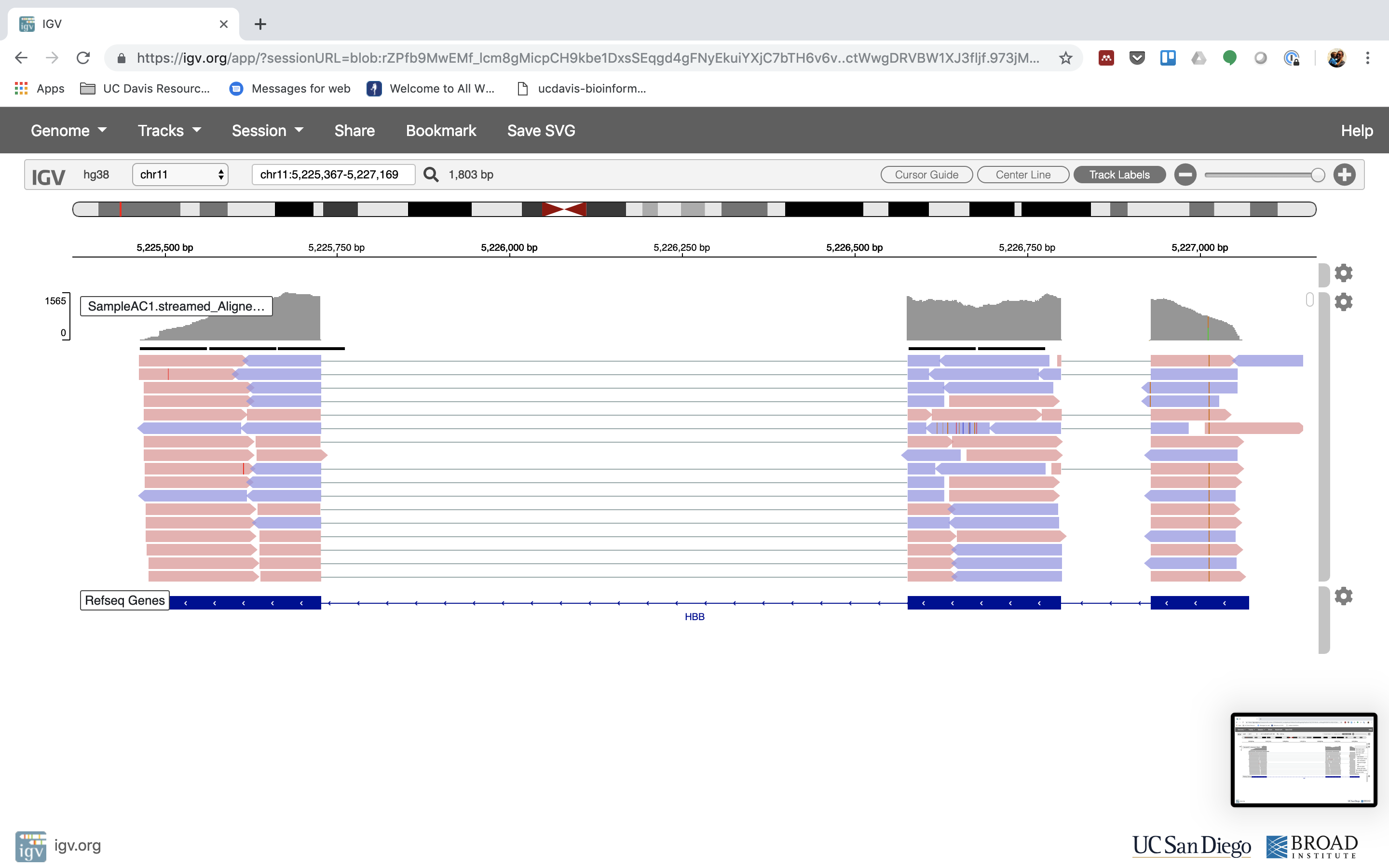
Here, we’ve used a reference transcriptome for Arabadopsis. However, one of the benefits of performing quantification directly on the transcriptome (rather than via the host genome), is that one can easily quantify assembled transcripts as well (obtained via software such as StringTie for organisms with a reference or Trinity for de novo RNA-seq experiments).
Next, we’re going to build an index on our transcriptome. The index is a structure that salmon uses to quasi-map RNA-seq reads during quantification. The index need only be constructed once per transcriptome, and it can then be reused to quantify many experiments. We use the index command of salmon to build our index:
There are a number of different options you can pass to the indexer to change its behavior (read more about those here), but the default should work well for most data.
Obtaining sequencing data
In addition to the index, salmon obviously requires the RNA-seq reads from the experiment to perform quantification. In this tutorial, we’ll be analyzing data from this 4-condition experiment [accession PRJDB2508]. You can use the following shell script to obtain the raw data and place the corresponding read files in the proper locations. Here, we’re simply placing all of the data in a directory called
data, and the left and right reads for each sample in a sub-directory labeled with that sample’s ID (i.e. DRR016125_1.fastq.gz and DRR016125_2.fastq.gz go in a folder called data/DRR016125).We’ll place these commands in a script called
dl_tut_reads.sh. To download the data, just run the script and wait for it to complete:Now might be a good time to grab a cup of coffee (or tea).
Quantifying the samples
Now that we have our index built and all of our data downloaded, we’re ready to quantify our samples. Since we’ll be running the same command on each sample, the simplest way to automate this process is, again, a simple shell script (
quant_tut_samples.sh): Pss cctv download for mac.This script simply loops through each sample and invokes
salmon using fairly barebone options. The -i argument tells salmon where to find the index -l A tells salmon that it should automatically determine the library type of the sequencing reads (e.g. stranded vs. unstranded etc.). The -1 and -2 arguments tell salmon where to find the left and right reads for this sample (notice, salmon will accept gzipped FASTQ files directly). Cubase pro 9 download for mac. Finally, the -p 8 argument tells salmon to make use of 8 threads and the -o argument specifies the directory where salmon’s quantification results sould be written. Salmon exposes many different options to the user that enable extra features or modify default behavior. However, the purpose and behavior of all of those options is beyond the scope of this introductory tutorial. You can read about salmon’s many options in the documentation.After the salmon commands finish running, you should have a directory named
quants, which will have a sub-directory for each sample. These sub-directories contain the quantification results of salmon, as well as a lot of other information salmon records about the sample and the run. The main output file (called quant.sf) is rather self-explanatory. For example, take a peek at the quantification file for sample DRR016125 in quants/DRR016125/quant.sf and you’ll see a simple TSV format file listing the name (Name) of each transcript, its length (Length), effective length (EffectiveLength) (more details on this in the documentation), and its abundance in terms of Transcripts Per Million (TPM) and estimated number of reads (NumReads) originating from this transcript.After quantification
That’s it! Quantifying your RNA-seq data with salmon is that simple (and fast).Once you have your quantification results you can use them for downstream analysis with differential expression tools like swish,DESeq2,edgeR,limma, orsleuth.Using the tximport package, you can import salmon’s transcript-level quantifications and optionally aggregate them to the gene level for gene-level differential expression analysis.You can read more about how to import salmon’s results into DESeq2 by reading the
tximport Auto tune voice changer download mac. sectionof the excellent DESeq2 vignette.For instructions on importing for use with edgeR or limma, see the tximport vignette.For using salmon with swish for transcript level analysis, see the swish vignette.For preparing salmon output for use with sleuth, see the wasabi package.- When you are building a salmon index, please do not build the index on the genome of the organism whose transcripts you want to quantify, this is almost certainly not want you want to do and will not provide you with meaningful results. ↩
We recommend using Java Web Start, but if you plan to use StarGenetics with limited internet connectivity please feel ree to download a version for your operating system.
How to start Linux version?
StarGenetics.bin file is bash shell script. When you execute it, it will unpack and start StarGenetics. StarGenetics requires Java Runtime to be installed on your system.
If you are on a MIT Athena workstation, download StarGenetics and then in Terminal type:
add -f java_v1.6.0_31
bash ~/Downloads/StarGenetics.bin
For more information about this issue visit IS&T's page about Athena and Java Web Start.
How to start Windows version?
StarGenetics.exe can be executed. StarGenetics requires Java Runtime to be installed on your system.
How to start MacOS X version?
Download Rna Star For Macbook
StarGenetics.zip should be decompressed. It will create a StarGenetics application that can be executed. It requires at least MacOSX 10.5. If you are on MacOS 10.8 or newer, please download Java Runtime from Oracle.
